Concierge Medical Services
A Blog for the Healthcare Web 2.0 Generation
June 25, 2015
The startup learning exponential at Medicast
June 24, 2013
Injected Nanoparticles Maintain Blood Sugar Levels In Diabetic Mice For Ten Days:
Written by Peter Murray
![[Source: Broad Institute]](http://singularityhub.com/wp-content/uploads/2013/05/image2A6.jpg)
[Source: Broad Institute]
While the exact cause of type 1 diabetes is unknown it is thought to be an autoimmune disorder in which the body attacks cells in the pancreas that produce insulin, a hormone that causes blood sugar to be taken up by cells and used for energy. In type 1 diabetes the pancreas makes little or no insulin, leading to high levels of sugar in the blood which can lead to blindness, limb amputation and kidney failure. Untreated, the disease is fatal.
Right now type 1 diabetes patients have to receive insulin shots, several times a day, for the rest of their lives to keep blood sugar levels under control. And there’s added risk if the patient’s dose isn’t correct – too much insulin is harmful too. The new device would relieve patients of the need to monitor and control their own blood sugar levels by controlling them automatically.
![Nano-networks of porous beads are engineered to respond to high levels of blood sugar by releasing insulin. [Source: Journal of Agriculture and Food Chemistry]](http://singularityhub.com/wp-content/uploads/2013/05/nano.jpg)
Nano-networks of porous beads are engineered to respond to high levels of blood sugar by releasing insulin. [Source: Journal of Agriculture and Food Chemistry]
“This technology effectively creates a ‘closed-loop’ system that mimics the activity of the pancreas in a healthy person, releasing insulin in response to glucose level changes,” Zhen Gu, lead author of the study and an assistant professor in the joint biomedical engineering program at North Carolina State and University of North Carolina Chapel Hill, said in a press release.
The study was published recently in the Journal of Agricultural and Food Chemistry. The next step, the researchers say, is to test the technology in humans.
The current technique works in principle similarly to the ‘Silicon Pancreas,’ a microchip that regulates blood sugar levels by mimicking both the insulin-producing beta cells and glucagon-producing alpha cells in the pancreas. The current technique is more elegant in its simplicity, however, as the Silicon Pancreas requires a separate blood sugar level monitor embedded in the skin.
Diabetes affects 366 million people worldwide. It’s the seventh leading cause of death in the United States where 25.8 million children and adults, or 8.3 percent of the population, have the disease. And as people continue to grow in size, so will diabetes as an epidemic, and devices such as the one developed by Dr. Zhen and colleagues will be needed more than ever.
Engineered molecules boost immune attack on cancer, researchers say
BY CHRISTOPHER VAUGHAN - Building on previous research showing that cancer cells send signals to the immune system to avoid being attacked, Stanford scientists have engineered new molecules that are highly proficient at neutralizing those signals.
Christopher Garcia

The molecules dramatically increase the effectiveness of certain existingcancer therapies and may open up other avenues for treating cancerusing patients' own immune systems.
School of Medicine researchers have previously shown that CD47, a molecule found on the surface of many cancers, acts as a "don't eat me" signal that protects the cancer from roving immune cells called macrophages. The scientists found that when they used drugs to block this "don't eat me" signal, macrophages engulfed and destroyed the cancer cells.
More recently, Stanford scientists wondered if they could engineer molecules that block the signal more effectively. They began by modifying a protein called SIRP-alpha, which is found on macrophages and is the natural receptor for CD47. In a paper published online May 30 in Science, a team of scientists working with professors Christopher Garcia, PhD, and Irving Weissman, MD, report that they have engineered new versions of SIRP-alpha that bind much more strongly to CD47 than the natural version of the molecule, making them extremely potent blockers of the "don't eat me" signal.
Two MD/PhD students in the Stanford Medical Scientist Training Program — Kipp Weiskopf, MPhil, and Aaron Ring, MS — devised the strategy for engineering these new molecules and were lead authors of the Science paper.
"This is the best example of how science and medical trainingcan lead to discovery and medical translation," said Weissman, professor of pathology and of developmental biology. "Weiskopf and Ring fleshed out the ideas that led to this project, bringing the complementary expertise of our groups together."
The engineered SIRP-alpha variants bind approximately 50,000 times more strongly to CD47 than native SIRP-alpha, making them highly potent drugs, Weiskopf said. But the researchers found blocking the CD47 "don't eat me" signal is not, by itself, enough to stimulate macrophages to attackcancer cells. Instead, the group demonstrated that blocking CD47 boosts the activity of macrophages when a second "eat me" signal is provided. This signal can be provided by certain anti-cancer antibody therapeutics, many of which are already approved by the FDA and in use clinically.
These findings have tremendous therapeutic implications, Ring said, because the high-affinity SIRP-alpha molecules could selectively boost the destruction of cancer cells by these antibody therapies, without increasing toxic side effects such as the destruction of healthy, non-cancerous cells.
The authors found that their engineered SIRP-alpha molecules would enhance a number of widely used antibody therapies such as rituximab (which targets certain lymphomas and leukemias), trastuzumab (which targets certain kinds of breastcancer) and cetuximab (which targets certain kinds of head and neck cancer and colon cancer).
When physicians used an anti-cancer antibody like these to fight cancer, the antibody's "eat me" signal to the macrophage is fighting against the cancer's "don't eat me" signal coming from the CD47, the researchers explain. When the high-affinity SIRP-alpha binds tightly to the CD47 molecules on the cancer, it takes the brakes off the macrophage attack on the cancercells.
"When physicians use an anti-cancer antibody like trastuzumab to fight breast cancer, the 'don't eat me signal' from CD47 on the cancer limits the effectiveness of the antibody," said Ring. "Potentially, any anti-cancer antibody that stimulates immune cells could benefit from this combination therapy."
In one experiment, when either rituximab or the high-affinity SIRP-alpha variants were used by themselves on mice with human lymphomas, they were only able to slow tumor growth. However, when the two therapies were combined, researchers found they were able to eliminate the tumors completely. "Antibodies are like guided missiles in terms of targeting cancercells," Weiskopf said. "By combining them with the high-affinity SIRP-alpha variants, you are adding high-explosive warheads to those missiles ."
The researchers hope that their work with high-affinity SIRP-alpha molecules will lead to more effective targeted-cancertherapies in the clinic. "This work further validates the strategy of targeting CD47 to enhance macrophage attack of cancer," according to Weissman. "This strategy must still be tested in humans to evaluate safety, but anti-CD47 antibodies are on schedule for the first trials in patients next year."
Other authors of the paper include graduate student Chia Chi M. Ho; instructor Jens-Peter Volkmer, MD; former postdoctoral scholar Aron Levin, PhD; postdoctoral scholars Anne Kathrin Volkmer, MD, Engin Özkan, PhD, and Nathaniel Fernhoff, PhD; and Matt van de Rijn, MD, PhD, professor of pathology.
This work was supported by the National Cancer Institute, theNational Institute of Diabetes and Digestive and Kidney Diseases, the Stanford Medical Scientist Training Program, the School of Medicine's SPARK program, the Deutsche Forschungsgemeinschaft, the Joseph & Laurie Lacob Gynecologic/Ovarian Cancer Fund, the Virginia and D.K. Ludwig Fund for Cancer Research and the Howard Hughes Medical Institute.
http://www.healthcanal.com/
http://www.healthcanal.com/
Melanoma now detectable by skin odor

SolarScan, a device to detect skin cancer, is demonstrated at its launch in Sydney May 7, 2002. The SolarScan works by capturing an image of a patient's skin spot and then an image analsis software compares the features against images of melanomas and non-melanomas in a database. Melanoma is the most common form of cancer in men and women aged 15-44 and is the most deadly form of skin cancer. Credit:Reuters
Responsible for more than 75 percent of skin cancer deaths, melanoma is a cancer of the skin, which affects the melanocytes (pigment carrying cells of the body). A new study now suggests that this dangerous cancer may be easily detectable - by odors from skin cells.
The study, carried out by a team of researchers from the Monell Center revealed two new ways to help early detection of skin cancer- by odors emitted by human skin, and by using a nanotechnology-based sensor that could differentiate between normal skin cells and melanoma skin cells.
"There is a potential wealth of information waiting to be extracted from examination of VOCs associated with various diseases, including cancers, genetic disorders, and viral or bacterial infections," senior author George Preti, PhD and an organic chemist at Monell, noted.The researchers took into consideration the fact that human skin produces a number of airborne chemical molecules, also known as volatile organic compounds (VOCs), many of which emit odor too, and put it to use as an early melanoma detection tool.
For this study, the researchers made use of sophisticated analytical and sampling techniques that helped detect VOCs emitted from melanoma skin cells at different levels of progression of the disease. They also sampled VOCs emitted from normal skin cells by using an absorbent device. Analysis of these VOCs was done using mass spectrometry and gas chromatography techniques.
To translate the results obtained from the VOC analysis into a diagnostic analysis, a reliable and portable sensor would be required, to which, the researchers used a nano-sensor.
The nano-sensor was made up of two nano-sized tubes that were coated with strands of DNA. The use of DNA as a sensor could be clarified by the fact that DNA tend to act as a wonderful sensor, which makes identification of the target easy. The results so obtained revealed a stunning fact- human skin cells emit odor different from that of melanoma skin cells; a new early diagnostic tool which may help cut down death rates due to melanoma considerably.
"This study demonstrates the usefulness of examining VOCs from diseases for rapid and noninvasive diagnostic purposes," Preti said. "The methodology should also allow us to differentiate stages of the disease process."
The study is now published in the Journal of Chromatography B.
Innovative nanoparticle therapy to treat infected burn wounds
(Nanowerk Spotlight) Despite significant advances in the medical/surgical management of severe thermal injury, wound infection and subsequent sepsis persist as frequent causes of morbidity and mortality for burn victims not only due to the extensive compromise of the protective barrier against microbial invasion, but also as a result of growing pathogen resistance to our therapeutic options.Burn wounds are further complicated by injury induced suppression of the immune system, further facilitating localized colonization and systemic entry of microbes from both endogenous and exogenous origins. Candida yeast species represent a prevalent threat to these patients, many variants demonstrating resistance to first line systemic antifungal treatments as these medications are not directly toxic, but instead inhibit yeast growth through a single mechanism of action.Therefore, innovative therapies are urgently needed that overcome mechanisms of pathogen resistance, and are easily administered without concerning systemic side effects.Nanoparticles represent a practical platform through which both established drugs and novel agents can be encapsulated and delivered more effectively or via new routes to target and reach pathogen cells/machinery with greater success then their macromolecular counterparts.In a paper published in the June 14, 2013 online edition of Nanomedicine: Nanotechnology, Biology and Medicine ("Amphotericin B releasing nanoparticle topical treatment of Candida spp. in the setting of a burn wound") we demonstrated that encapsulating Amphotericin B, a intravenously administered potent fungicidal polyene macrolide, in nanoparticles increased its killing impact against numerous candida species, was more effective at preventing candidal biofilm formation, and cleared a mouse burn model infected with candida more effectively than solubilized amphotericin.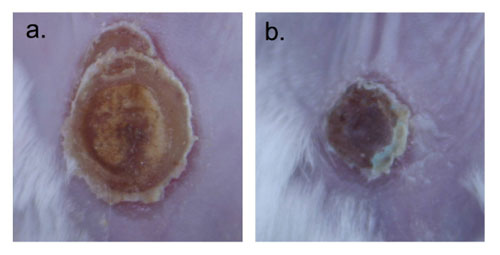 Images were taken on day 9 after infection. a) is topically applied solubilized Amphotericin B and b) Amphotericin B nanoparticle applied. (Images: Dr. Friedman, Montefiore - Albert Einstein College of Medicine)Materials at the nanoscale have a greater likelihood of interacting with their desired target, here, the cell wall, and therefore the payloads have greater efficacy. Furthermore, the nanoparticles themselves can physically damage pathogen structures and prevent cell-to-cell communication and sharing of resources, offering synergists but different antifungal properties.Encapsulating amphotericin in nanoparticles not only made it more effective, but also allowed for it to be directly and topically applied to a wound – an option not currently available. Systemic delivery, while very effective, comes at a great cost as amphotericin has multiple well established side effects and toxicities associated with its use.Our team here at Einstein represents a broad range of expertise calling from multiple departments including medicine (infectious diseases, dermatology), physiology and biophysics, and immunology and microbiology, highlighting the multidisciplinary nature of nanotechnology research. Other authors included David Sanchez; David Schairer; Chaim Tuckman-Vernon; Jason Chouake; Allison Kutner; Joy Makdisi; Joel Friedman; and Joshua d Nosanchuk.By Dr. Adam Friedman, Assistant Professor of Medicine (Dermatology)/ Physiology and Biophysics, and Director of Dermatologic Research at Montefiore Medical Center - Albert Einstein College of Medicine.
Images were taken on day 9 after infection. a) is topically applied solubilized Amphotericin B and b) Amphotericin B nanoparticle applied. (Images: Dr. Friedman, Montefiore - Albert Einstein College of Medicine)Materials at the nanoscale have a greater likelihood of interacting with their desired target, here, the cell wall, and therefore the payloads have greater efficacy. Furthermore, the nanoparticles themselves can physically damage pathogen structures and prevent cell-to-cell communication and sharing of resources, offering synergists but different antifungal properties.Encapsulating amphotericin in nanoparticles not only made it more effective, but also allowed for it to be directly and topically applied to a wound – an option not currently available. Systemic delivery, while very effective, comes at a great cost as amphotericin has multiple well established side effects and toxicities associated with its use.Our team here at Einstein represents a broad range of expertise calling from multiple departments including medicine (infectious diseases, dermatology), physiology and biophysics, and immunology and microbiology, highlighting the multidisciplinary nature of nanotechnology research. Other authors included David Sanchez; David Schairer; Chaim Tuckman-Vernon; Jason Chouake; Allison Kutner; Joy Makdisi; Joel Friedman; and Joshua d Nosanchuk.By Dr. Adam Friedman, Assistant Professor of Medicine (Dermatology)/ Physiology and Biophysics, and Director of Dermatologic Research at Montefiore Medical Center - Albert Einstein College of Medicine.
Read more: http://www.nanowerk.com/spotlight/spotid=30997.php#ixzz2X9QjwHzd
 Images were taken on day 9 after infection. a) is topically applied solubilized Amphotericin B and b) Amphotericin B nanoparticle applied. (Images: Dr. Friedman, Montefiore - Albert Einstein College of Medicine)Materials at the nanoscale have a greater likelihood of interacting with their desired target, here, the cell wall, and therefore the payloads have greater efficacy. Furthermore, the nanoparticles themselves can physically damage pathogen structures and prevent cell-to-cell communication and sharing of resources, offering synergists but different antifungal properties.Encapsulating amphotericin in nanoparticles not only made it more effective, but also allowed for it to be directly and topically applied to a wound – an option not currently available. Systemic delivery, while very effective, comes at a great cost as amphotericin has multiple well established side effects and toxicities associated with its use.Our team here at Einstein represents a broad range of expertise calling from multiple departments including medicine (infectious diseases, dermatology), physiology and biophysics, and immunology and microbiology, highlighting the multidisciplinary nature of nanotechnology research. Other authors included David Sanchez; David Schairer; Chaim Tuckman-Vernon; Jason Chouake; Allison Kutner; Joy Makdisi; Joel Friedman; and Joshua d Nosanchuk.By Dr. Adam Friedman, Assistant Professor of Medicine (Dermatology)/ Physiology and Biophysics, and Director of Dermatologic Research at Montefiore Medical Center - Albert Einstein College of Medicine.
Images were taken on day 9 after infection. a) is topically applied solubilized Amphotericin B and b) Amphotericin B nanoparticle applied. (Images: Dr. Friedman, Montefiore - Albert Einstein College of Medicine)Materials at the nanoscale have a greater likelihood of interacting with their desired target, here, the cell wall, and therefore the payloads have greater efficacy. Furthermore, the nanoparticles themselves can physically damage pathogen structures and prevent cell-to-cell communication and sharing of resources, offering synergists but different antifungal properties.Encapsulating amphotericin in nanoparticles not only made it more effective, but also allowed for it to be directly and topically applied to a wound – an option not currently available. Systemic delivery, while very effective, comes at a great cost as amphotericin has multiple well established side effects and toxicities associated with its use.Our team here at Einstein represents a broad range of expertise calling from multiple departments including medicine (infectious diseases, dermatology), physiology and biophysics, and immunology and microbiology, highlighting the multidisciplinary nature of nanotechnology research. Other authors included David Sanchez; David Schairer; Chaim Tuckman-Vernon; Jason Chouake; Allison Kutner; Joy Makdisi; Joel Friedman; and Joshua d Nosanchuk.By Dr. Adam Friedman, Assistant Professor of Medicine (Dermatology)/ Physiology and Biophysics, and Director of Dermatologic Research at Montefiore Medical Center - Albert Einstein College of Medicine.Read more: http://www.nanowerk.com/spotlight/spotid=30997.php#ixzz2X9QjwHzd
April 12, 2013
Regenerative Heart Medicine Could Get Boost With Nanotechnology
Researchers at the Stanford University School of Medicine have developed a new visualization technique which they believe could eventually help make the repair of damaged hearts through regenerative medicine a reality.
In a study published in Wednesday’s edition of the journal Science Translational Medicine, senior author and Stanford radiology professor Sam Gambhir and colleagues describe how they plan to mark the stem cells which would be used in the repair process.
By marking the cells, doctors would be able to track them by using standard ultrasounds as they leave the needle and enter a patient’s body. The process would allow for the stem cells to be guided to their intended destination more precisely, and would also allow doctors to monitor them using magnetic-resonance imaging (MRI) technology for several weeks afterwards, the researchers explained.
To date, both human and animal trials in which stem cells were injected into cardiac tissue to treat severe heart attacks or heart failure have been largely unsuccessful, said Gambhir.
“We’re arguing that the failure is at least partly due to faulty initial placement,” he explained in a statement. “You can use ultrasound to visualize the needle through which you deliver stem cells to the heart. But once those cells leave the needle, you’ve lost track of them.”
For this reason, scientists have been unable to precisely determine whether or not the stem cells actually reached the heart wall, and whether they remained there or diffused away from the cardiac tissue. In addition, there has been no way to determine how long the cells managed to stay alive, or if they successfully replicate and eventually develop into heart cells.
Gambhir’s team method could help answer some of those questions.
“All stem cell researchers want to get the cells to the target site, but up until now they’ve had to shoot blindly,” he said. “With this new technology, they wouldn’t have to. For the first time, they would be able to observe in real time exactly where the stem cells they’ve injected are going and monitor them afterward.”
“If you inject stem cells into a person and don’t see improvement, this technique could help you figure out why and tweak your approach to make the therapy better,” Gambhir added.
In addition to the issues surrounding the initial position of the therapeutic stem cells, tracking them once they enter the body has proven troublesome since there is no way to distinguish them from any other cell in the patient’s body. Since they normally cannot be tracked upon entering the body, if the attempt to repair the heart fails, doctors often are unable to pinpoint exactly why the process proved unsuccessful.
The new technique, however, aims to solve those problems by using extremely small nanoparticles that act as imaging agents. The nanoparticles, which have a diameter slightly less than one-third of a micron (or less that one-thirtieth the diameter of a red blood cell), are made of silica so that they can be visualized by ultrasound. Furthermore, an MRI contrast agent known as gadolinium was also added to the imaging agents.
Gambhir and his colleagues were able to successfully demonstrate that mesenchymal stem cells – a class of cells frequently used in heart-regeneration research – could store the nanoparticles without sacrificing any of their ability to survive, replicate and differentiate into living heart cells.
Lead author Jesse Jokerst, a postdoctoral scholar in Gambhir’s lab, said there were concerns that the signal would be fairly weak. However, he and his colleagues found that once they were ingested, they clumped together within the cells, reflecting the ultrasound waves far more dramatically and providing a far stronger signal than anticipated.
Despite the optimism, it will probably be at least three years before the technique can be tested in humans.
April 10, 2013
One Drug to Shrink All Tumors
A single drug can shrink or cure human breast, ovary, colon, bladder, brain, liver, and prostate tumors that have been transplanted into mice, researchers have found. The treatment, an antibody that blocks a "do not eat" signal normally displayed on tumor cells, coaxes the immune system to destroy the cancer cells.
A decade ago, biologist Irving Weissman of the Stanford University School of Medicine in Palo Alto, California, discovered that leukemia cells produce higher levels of a protein called CD47 than do healthy cells. CD47, he and other scientists found, is also displayed on healthy blood cells; it's a marker that blocks the immune system from destroying them as they circulate. Cancers take advantage of this flag to trick the immune system into ignoring them. In the past few years, Weissman's lab showed that blocking CD47 with an antibody cured some cases of lymphomas and leukemias in mice by stimulating the immune system to recognize the cancer cells as invaders. Now, he and colleagues have shown that the CD47-blocking antibody may have a far wider impact than just blood cancers.
"What we've shown is that CD47 isn't just important on leukemias and lymphomas," says Weissman. "It's on every single human primary tumor that we tested." Moreover, Weissman's lab found that cancer cells always had higher levels of CD47 than did healthy cells. How much CD47 a tumor made could predict the survival odds of a patient.
To determine whether blocking CD47 was beneficial, the scientists exposed tumor cells to macrophages, a type of immune cell, and anti-CD47 molecules in petri dishes. Without the drug, the macrophages ignored the cancerous cells. But when the anti-CD47 was present, the macrophages engulfed and destroyed cancer cells from all tumor types.
Next, the team transplanted human tumors into the feet of mice, where tumors can be easily monitored. When they treated the rodents with anti-CD47, the tumors shrank and did not spread to the rest of the body. In mice given human bladder cancer tumors, for example, 10 of 10 untreated mice had cancer that spread to their lymph nodes. Only one of 10 mice treated with anti-CD47 had a lymph node with signs of cancer. Moreover, the implanted tumor often got smaller after treatment—colon cancers transplanted into the mice shrank to less than one-third of their original size, on average. And in five mice with breast cancer tumors, anti-CD47 eliminated all signs of the cancer cells, and the animals remained cancer-free 4 months after the treatment stopped.
"We showed that even after the tumor has taken hold, the antibody can either cure the tumor or slow its growth and prevent metastasis," says Weissman.
Although macrophages also attacked blood cells expressing CD47 when mice were given the antibody, the researchers found that the decrease in blood cells was short-lived; the animals turned up production of new blood cells to replace those they lost from the treatment, the team reports online today in the Proceedings of the National Academy of Sciences.
Cancer researcher Tyler Jacks of the Massachusetts Institute of Technology in Cambridge says that although the new study is promising, more research is needed to see whether the results hold true in humans. "The microenvironment of a real tumor is quite a bit more complicated than the microenvironment of a transplanted tumor," he notes, "and it's possible that a real tumor has additional immune suppressing effects."
Another important question, Jacks says, is how CD47 antibodies would complement existing treatments. "In what ways might they work together and in what ways might they be antagonistic?" Using anti-CD47 in addition to chemotherapy, for example, could be counterproductive if the stress from chemotherapy causes normal cells to produce more CD47 than usual.
Weissman's team has received a $20 million grant from the California Institute for Regenerative Medicine to move the findings from mouse studies to human safety tests. "We have enough data already," says Weissman, "that I can say I'm confident that this will move to phase I human trials."
*Correction, 2 April 2013: One reference to the compound used to treat mice was previously named as CD47, but in all cases was the antibody to that protein, anti-CD47.
Subscribe to:
Posts (Atom)





